- Find our latest Case Studies
- Our Platforms
Our Platforms
- Platform 1: Bioactives Library – Shaping novel antimicrobial products
- Platform 2: Hits to Leads – Developing new innovative antibiotics
- Platform 3: Organoid modelling – Enhancing drug discovery
- Platform 4: Advanced AMR Modelling
- Platform 5: Human Challenge – Small scale clinical trials
- Platform 6: Diagnostics
- Platform 7: Randomised Control Trials
- Platform 8: Mapping and Modelling
- Platform 9: Nanotherapeutics
- Platform 10: Antibody Humanisation
- Platform 11: Long-Acting Therapeutics
Landmark pnuemococcal vaccine trial reaches key milestone
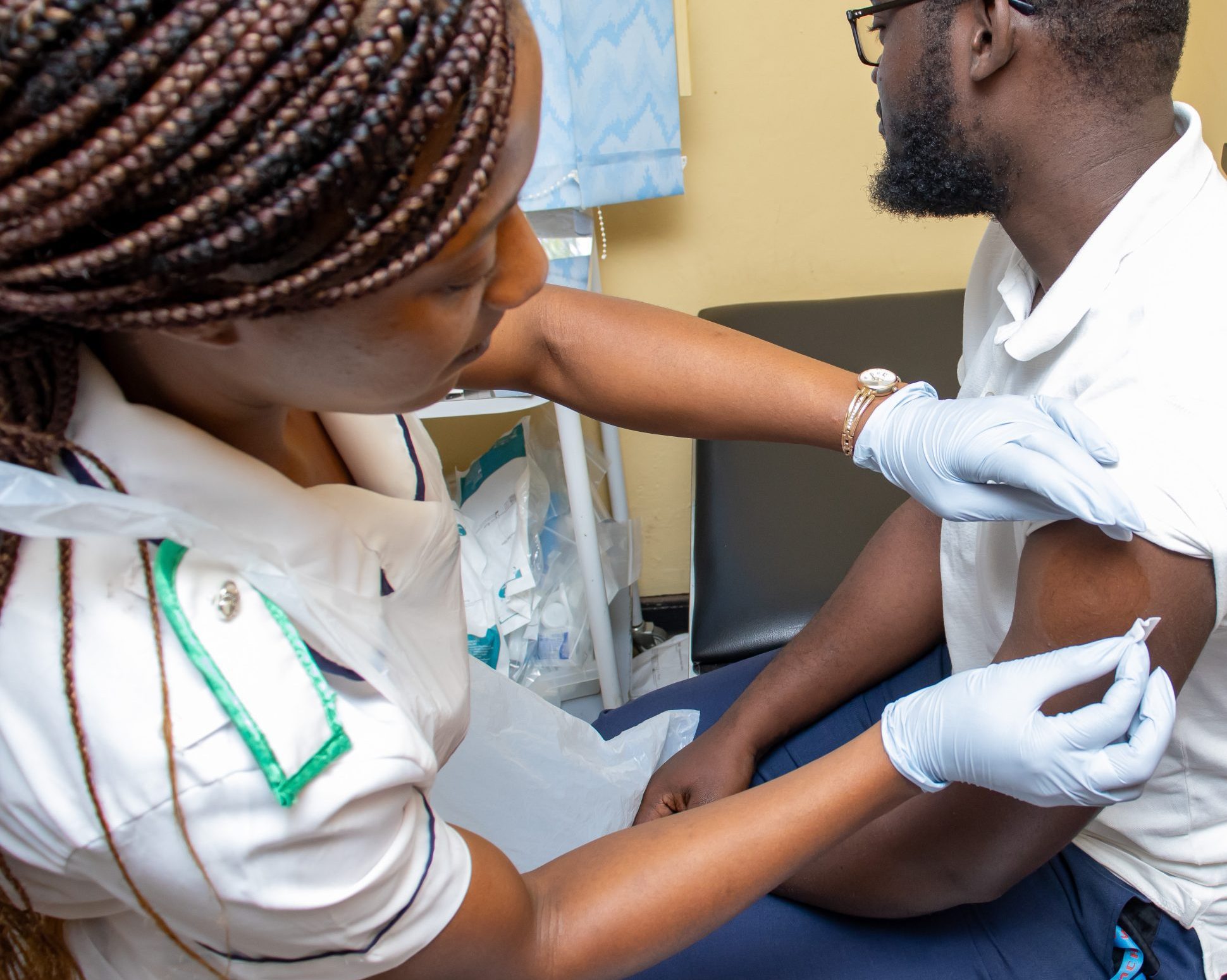
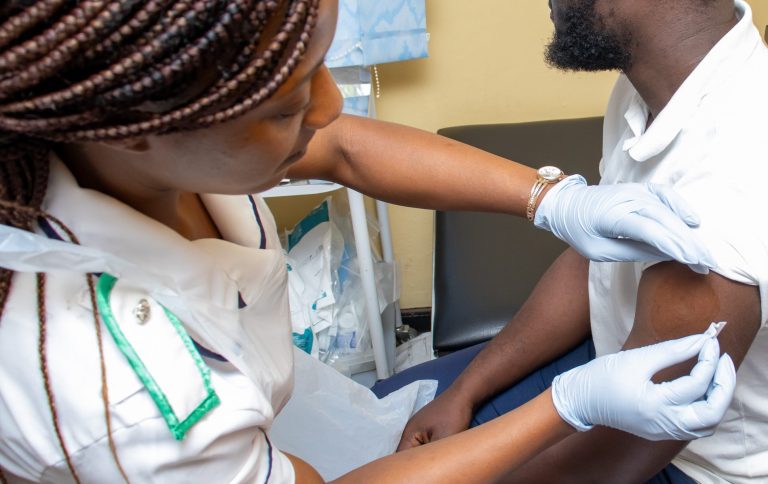
The Infection Innovation Consortium and partners have reached a significant milestone in a landmark clinical trial being delivered in Malawi to develop a novel vaccine for pneumococcal disease.
The trial is the first ever controlled human infection model (CHIM) programme to test a vaccine for efficacy against pneumococcus in Africa. It is part of a £3.2 million Medical Research Council (MRC) funded project being delivered by iiCON to bring forward a new vaccine, PnuBioVax, developed by UK SME ImmBio.
Over 100 people have now been enrolled for the trial, which is being led by the Malawi Liverpool Wellcome Research Programme. The trial will enrol 416 people in total over the course of 2026 – making it the biggest human challenge trial conducted in the country to date.
Addressing the global pneumococcal burden
Invasive Pneumococcal disease has high rates of antibiotic resistance and is a leading global cause of deaths in children, with most deaths occurring in low- and middle-income countries (LMICs).
Streptococcus pneumoniae, the bacteria responsible for pneumococcal disease has nearly 100 different strains, including increasingly antimicrobial-resistant strains such as Serotype 3 pneumococcus (SPN3).
People living in Malawi face a particular threat from SPN3. This strain, which causes invasive disease, is increasingly prevalent and has high rates of antimicrobial resistance. It poses a significant public health concern to the community, particularly children and individuals with HIV.
The dangers posed by SPN3 are not isolated to the Malawian population – this strain is also the most common cause of severe pneumonia in children in Europe.
However, the PnuBioVax vaccine targets proteins which are common across all pneumococcus serotypes, including SPN3, so there is strong hope that it could lead to a more holistic control response across many different strains of the disease.
Delivering Africa’s first Pneumococcal CHIM trial
The trial underway in Malawi is testing the PnuBioVax vaccine on young healthy adults under a controlled human infection model (CHIM) being led by the Malawi Liverpool Wellcome Research Programme.
The sample group is being vaccinated and then inoculated in their noses to understand if they are carrying a strain of the disease or not. This is then compared to the existing relevant vaccine and a placebo.
CHIM trials for pneumococcal disease have been well established at Liverpool School of Tropical Medicine (LSTM) for many years and this trial will build on the current £4.5m MARVELS (Malawi Accelerated Research in Vaccines by Experimental and Laboratory Systems) programme at the Malawi Liverpool Wellcome Research Programme.
Looking ahead
Following the trial, the vaccine could have the potential to be taken forward at scale by an industry partner which could develop and deliver a low-cost vaccine at scale – with potential for manufacturing and distribution across Africa. iiCON and ImmBio are now working to identify potential commercial partners who may support the next stage of development following the successful completion of the trial.
Professor Stephen Gordon, Director of Experimental Medicine at iiCON, is leading the programme. He said: “It’s great to see the team at MLW making such strong strides in the delivery of this important trial, which will help us to assess the impact of this potentially game-changing vaccine, on a local population.
“Reaching 100 participants is such a relatively short period of time is a fantastic achievement and sets us in good stead to complete the delivery of this important trial on time and to budget.
“This programme of activity is a great example of the collaborative global innovation that iiCON was designed to enable. We have had the opportunity to work alongside world-leading international partners to support a UK SME with a potential solution to a global health crisis, while addressing a key public health challenge in Malawi.
“This activity is also turning the dial for infectious disease capacity in Malawi; helping to deepen in-country capability and move us closer to a position where low-income countries are able to play a key role in developing and potentially even manufacturing their own locally responsive vaccines.”
Professor Henry Mwandumba, Director of the Malawi Liverpool Wellcome Research Programme, said: “This work has the potential to significantly improve health in Malawi and the region if the vaccine safely reduces pneumococcal carriage and transmission. Halting the spread of strains like SPN3 will help build healthier communities and advance MLW’s mission to deliver impactful health research.”